Welcome to the CTU Network’s Spring 2026 newsletter.
You can find out more about some of our recent activities below.
In this issue
News from around the Network
- Programme Manager’s Update
- New Thematic Groups Established
- New Guidance and Information for CTUs
- CTUs in the News
- Promoting Registered CTUs & the Network on Social Media
- Training for Chief Investigators
- The DISTINCT project
- Operations Group Updates
- Training & Development Opportunities
- Job Opportunities
Updates from collaborators & stakeholders

Programme Manager’s Update
As we move into spring, we’re pleased to share the latest updates from across the Network. Over the past few months, everyone has shown incredible commitment and collaboration, with new thematic groups beginning their work, important guidance being released from Ops group, and a wide range of activities taking shape across our Clinical Trials Units.
I’m delighted to let you know that the Chief Investigator Network Group (CING) led by Rustam Al-Shahi Salman have recently been made a permanent group with their transition from a task and finish group to an Operations Group, find out more on this and what DISOG, TM, QA, Monitoring, and Stats are up to in the Ops Group update (including a new publication from the TM group!).
It’s always great to see how much progress is being made through working collectively, such as responding to evolving regulatory frameworks and expanding training opportunities. It’s genuinely inspiring to see everyone support each other.
Thank you to all who contributed to the initiatives featured in this newsletter, and I hope you enjoy reading these updates on exciting projects and plans for upcoming events in the months ahead.
Best wishes,
Helen Evans
UKCRC Registered CTU Network Programme Manager

New Thematic Groups Established
The Network has established five new thematic groups to help deliver the priorities set out in our 2025–2030 strategy. These cover Artificial Intelligence, Leadership Development, Methods into Practice, Research Inclusion and Sustainability.
Following a strong response to the nominations process, membership has now been confirmed, and each group has identified its chair(s). All five groups have held their first meetings to begin scoping their activities and draft their Terms of Reference.
We look forward to sharing updates on their work in the next newsletter.
For details on each group’s remit and membership, please click here.

New Guidance and Information for CTUs
We are exceptionally pleased to announce the release of a new report by the Trial Management Operations Group.
We are very grateful to everyone involved in the production of this piece of work.
How do Registered Clinical Trials Units (CTUs) manage the delivery of funded trials?
In 2025, the Trial Management Group developed a survey to quantify how registered CTUs are managing their portfolio of funded trials in terms of preparation, time periods within the funding for set-up and delivery, and the ultimate delivery in their trials meeting award timelines.
This survey recorded for the first time the amount of unfunded work Registered CTUs undertake in funded trials. The findings have now been written up and can be viewed here.

CTUs in the News
If we’ve missed a mention of your CTU or one of your trials, please let us know on regctus@leeds.ac.uk.
Oxford Vaccine Group (Oxford Collaborative CTU)
Professor Sir Andrew Pollard, Director of the Oxford Vaccine Group, spoke to BBC Radio 4’s Today Programme in January 2026, following the announcement of the introduction of the MMRV vaccine into the Childhood UK immunisation schedule. Listen to the clip here.
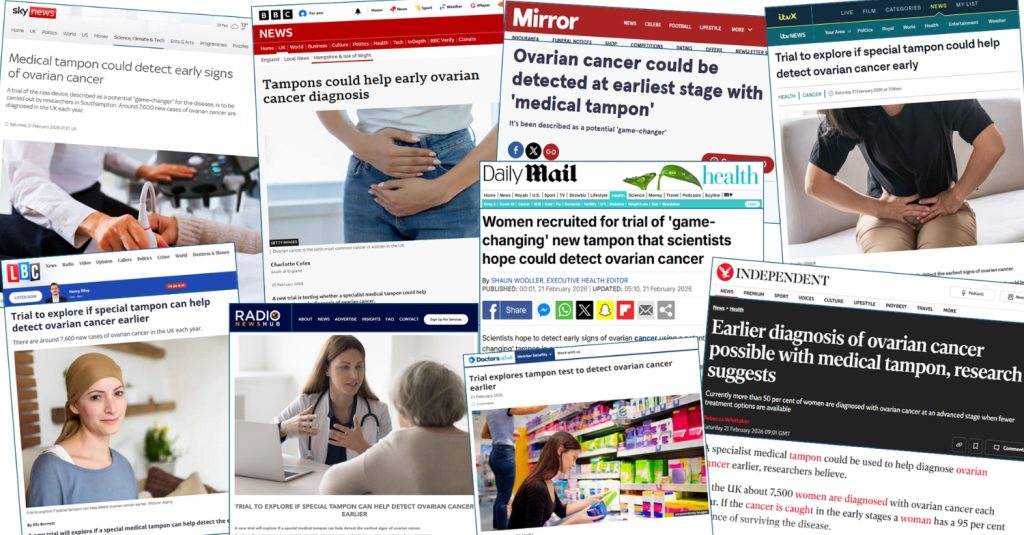
Southampton CTU
Did you see the coverage of the VIOLET study in the media in February 2026?
This trial aims to improve early diagnosis of Ovarian cancer using a diagnostic tampon and other patient samples to identify biomarkers, or genetic clues, that could indicate if cancer may be developing.
Find out more about the VIOLET study here.

Promoting Registered CTUs & the Network on Social Media
Since the beginning of the year, we’ve been using the Network’s website and social media channels to highlight the importance of the work of Registered CTUs and CTU staff using key dates in the calendar as inspiration. You can find out more about this below.
Over the next few months we’ll be recognising Earth Day (April), Dementia Action Week and International Clinical Trials Day (May), and Diabetes Awareness Week (June). If you’d like to get involved in any of these activities, please email regctus@leeds.ac.uk.
World Cancer Day – 4 February 2026
On World Cancer Day, we acknowledged the researchers, clinicians and organisations working together to test new ideas and build the evidence needed to improve cancer care.
That day also gave us an opportunity highlight some of the research that could make cancer treatment more effective and less difficult for patients. Many thanks to the CTUs who submitted their trials for inclusion.
Read our news piece here.
International Day of Women and Girls in Science – 11 February 2026
This day celebrates the vital contribution women make to science and research, while also recognising the ongoing gender inequalities that continue to hold back progress.
We used the occasion to spotlight women’s leadership across the Network.
You can find out more here.
Rare Disease Day – 28 February 2026
Thousands of rare diseases collectively affect millions of people in the UK. Because each individual condition is uncommon, conducting clinical trials to assess new treatments can be challenging. Sadly, this means many rare diseases lack effective treatment with no research ongoing to give patients hope.
The day gave us space to talk about the importance of the Registered CTU model in taking forward this important research, as well as share information on a number of rare disease trials being undertaken across the Network.
Many thanks to Prof. Max Parmar for his thoughtful blog piece and to the CTUs who submitted their trials for inclusion.

International Women’s Day – 8 March 2026
For International Women’s Day, we highlighted a range of women’s health initiatives led by Registered CTUs and celebrated some of the individual researchers across the Network. Thanks to all the CTUs who nominated their trials and staff members.
On 17 March 2026, we also hosted “Women at the helm: reflections on leadership, over changing times” featuring Prof. Julia Brown and Prof. Judith Bliss, in conversation with Prof. Kerry Hood. A recording of the event is available on the Network’s Vimeo channel. Contact the Secretariat at regctus@leeds.ac.uk for details.

Training for Chief Investigators
The next ‘How to be a Great Chief Investigator for Clinical Trials’ workshop is scheduled to take place on 5 May 2026 and will be hosted by Nottingham CTU. Please share these details with any current or new CIs who might be interested in further developing their practice.
As with previous events, this workshop has been awarded 5 external (category 1) CPD credits by the Federation of the Royal College of Physicians.
The workshop is designed to equip researchers with the skills and knowledge necessary to excel as Chief Investigators, through:
~ Expert guidance from experienced Chief Investigators with a diverse range of backgrounds and specialties.
~ The opportunity to meet and gain insights from specialists at Registered CTUs, including statisticians and trial managers.
~ Interactive activities to develop practical understanding of the issues.
~ A collaborative learning environment that fosters peer support.
A key feature of the workshop is the opportunity for delegates to develop their own peer-support network, enabling ongoing collaboration and shared learning beyond the event.
Further information and a registration link can be found here.


The DISTINCT project
There is increasing recognition of the need to make sure health research is inclusive. Collecting demographic information from participants helps monitor equal access to research and make sure results are relevant to people from all backgrounds, however there is no UK standard for asking research participants about their demographic characteristics.
We wanted to address this gap, and this is how the DISTINCT project was born. DISTINCT is a collaboration between the Cancer Prevention Trials Unit at Queen Mary University of London and The Institute of Cancer Research’s Clinical Trials and Statistics Unit. Our aim is to produce a set of demographic questions to improve monitoring of research inclusivity.

In 2024, we did a public consultation exercise with 29 patient and public representatives. We gathered feedback on potential questions, focusing on protected characteristics defined under the UK’s Equality Act (2010) and potentially underserved groups identified by the NIHR’s INCLUDE project.
The project team, including two PPI advisors, selected questions from surveys conducted by UK and devolved nation governments, to allow comparison with national datasets. We asked the public for their views on the questions, instructional text, data storage and confidentiality. A final question set was collated, containing 15 questions covering 12 characteristics: Age, Sex, Gender, Trans status, Sexual orientation, Ethnicity, Religion, Language, Education, Employment status, Disability and long‑term health conditions and Caring responsibilities. Further information can be found in our recent publication.
The DISTINCT question set is now being piloted and is available to all non-commercial UK researchers who agree to provide feedback on its implementation and acceptability. To date we have received 27 registrations for the pilot. Fourteen different trials units are using DISTINCT across different diseases including cancer, Parkinson’s and lung disease. We have also received registrations for other types of projects including PPI and behavioural science.
We are currently gathering feedback on the implementation of the question set so far. We are also planning a wider evaluation to assess the usefulness and acceptability of the question set, where we will gather aggregate data on question set completeness and use the results to refine the questions. We will be presenting DISTINCT at a UKCRC CTU webinar on 23rd April – please do join us then to find out more. Alternatively if you would like to take part in the pilot, or would like any other information, please email us at DISTINCT@icr.ac.uk or visit our webpage.
PPI reimbursement was funded by a Queen Mary University of London (QMUL) Centre for Public Engagement Small Grant Award, with supplementary funding from: Cancer Research UK (CRUK) Clinical Trials Unit grant to the Clinical Trials and Statistics Unit at the Institute of Cancer Research (CTUQQR-Dec22/100004), CRUK QMUL Cancer Prevention Trials Unit, QMUL Pragmatic Clinical Trials Unit and QMUL Barts Clinical Trials Unit. The implementation phase of the pilot is funded by Cancer Research UK: Clinical Trial Unit EDI Funding (CTUEFR-May25/100001).

QA Group Update
Supporting CTUs with the New Regulations
The priority for the QA Group in early 2026 has been helping Network CTUs prepare for and understand the new clinical trials regulations, including the associated regulatory guidance.
A key part of this is the ongoing work to develop guidance for CTUs on how to interpret and implement the annexes to ICH Good Clinical Practice Guideline E6(R3) in a proportionate manner. A QA Clinic online meeting was held on 9th March 2026 to discuss this project and what its final output might look like. Input on ICH GCP annex 1 has been sought from other Network groups, and the draft guidance will be shared with MHRA representatives for review.
Feedback on the First Year of QA Clinics
We ran a survey to gather feedback from Network CTUs on the first year of QA Clinics in preparation for planning the schedule for the rest of 2026. The comments we receive suggests these online sessions have been valuable and facilitate cross-CTU collaboration and networking outside of the national meeting. Thank you to everyone who took the time to share their thoughts.
Launching the CTU Knowledge-Sharing Repository
We have recently launched a repository (for member CTUs only) to share information and documents. This has huge potential benefits for CTUs to share learning and avoid reinventing any wheels, and highlights the value of the Network in efficient and effective clinical trial conduct. All CTU QA representatives should now have access. If you don’t, please contact the Secretariat on regctus@leeds.ac.uk.
Group Membership
We’re pleased to welcome Kelly Gee (Centre for Trials Research, Cardiff) to the QA Operations Group, and we’d like to thank everyone who expressed an interest in joining. We’re also delighted to confirm that Emily Swinburne (Newcastle Clinical Trials Unit) has been appointed as the new Deputy Chair.
– Will Cragg (Leeds Clinical Trials Research Unit)
Data & IS Group (DISOG) Update
Data & IS National Meeting 2026
The 2025 Data & IS National Meeting already feels like a distant memory. However, it was a very successful event with an overwhelming response from nearly 60 representatives across 38 UKCRC-registered CTUs, and a 100% ‘excellent’ rating. We are hoping to carry that momentum into the next national meeting. Planning is now underway for September 2026.
One of our priorities this year is to strengthen informal networking opportunities, which members consistently tell us they value. We also plan to invite vendors again, following another successful showing last year. Watch this space for specific venue and registration details.
Data Management Guidance
We remain committed to providing academic CTU focused DM guidance. Following the publication of the ‘Data Cleaning & Querying’ and ‘Data Reporting’ documents, we are in the final stages of preparing the ‘CRF/eCRF Development’ and ‘Study Database Lifecycle’ guidance for release.
Looking forward, we are currently planning a new CDISC Working Group. This will run alongside our existing active working groups, including the REDCap Validation group, which continues to develop community-led documentation. If you are interested in joining the CDISC Working Group or wish to champion any additional groups, please get in touch with the UKCRC Secretariat at regctus@leeds.ac.uk.
Upcoming Work and Suggested Topics
Plans for 2026 are informed by feedback from across our community. We are currently planning a webinar on Risk Assessment. We are also conducting a survey regarding MedDRA following recent significant regulatory changes, to better understand medical coding methods across Clinical Trials Units. Our goal is to investigate how CTUs are navigating these changes, identify which guidance is being used across the network, and highlight any existing gaps. If the survey identifies a need a working group will be established to produce formal guidance. If your CTU hasn’t already completed the survey, please do so by 31st March 2026
DISOG Membership Update
We are pleased to announce that Karen Boardman (Exeter CTU) and Naomi Campton (Birmingham CTU) have joined the group. We very much look forward to working with them. A big thank you to everyone who applied.
– Amanda Loban (Sheffield CTRU), John Turgoose (Hull Health Trials Unit), & Lindsey Masters (UCL Innovative CTU)
Trial Management Group Update
National Trial Managers’ Meeting 2026
Planning is underway for the next National Meeting, which will take place online in November. A save‑the‑date is expected to go out to members in March. The group is now starting to shape the agenda for the day. If you’re interested in sharing your work at the event, or if you have suggestions for topics you’d like to see included, please get in touch with the Secretariat at regctus@leeds.ac.uk.
New Report on Trial Set-up Activities Published
The survey circulated last year to explore the work carried out by units between funding being awarded and the official grant start date has now been written up. You can read the survey results here. We hope this will help demonstrate the amount of unfunded work taking place across the Network. Thank you to everyone who shared their experiences.
Potential Trial Repository for Participant Information Sheets
A short survey will be sent out soon via the Trial Managers JISCMail list units whether a shared Participant Information Sheet Library would be a useful resource for the Network. If you are a TM representative, please keep an eye out for this.
Guidance on working with Private Hospitals
The group is continuing work on high-level guidance to support including private hospitals in publicly funded trials, whether as full sites or for specific trial activities. More information will be shared in due course.
New actions
Work has begun on a project looking at the tools and checklists used during both grant development and delivery. A second project is starting to explore trial websites. More details on these new activities will follow in the next newsletter.
– Vicki Barber (Oxford Clinical Trials Research Unit) & Gina Cranswick (Edinburgh CTU)
Statistics Group Update
Webinar Series
Our popular webinar series continues with a broad range of topics from expert speakers. Upcoming talks will address incorporating historical information in trials and designing follow-up trials. Further webinars are also planned on the use of AI in trials and the ethical challenges this raises for clinical trial statisticians.
Activities
The Statistics group is currently involved in several activities including use of AI in CTUs, a survey to garner CTU experiences in collaborating with industry from a statistical perspective and providing feedback to the QA Group on the new ICH GCP E6(R3) guideline implemented in 2025.
We are also developing ideas for new projects in response to the 2025-2030 UKCRC CTU Strategy and Workplan, such as producing a guidance paper for statisticians working on decentralised trials and linking in with the newly established AI in Trials thematic group.
Suggestions for projects the Statistics group could focus on are always welcome; please email these to the Secretariat on regctus@leeds.ac.uk.
Induction and training materials for new statisticians
A task group has combined induction checklists from 10 CTUs into a single document that units can use to record on‑the‑job training and experience for new trial statisticians. A draft was shared for comment at the national meeting in Oxford in June 2025 and has since been circulated for piloting. We also plan to build on this work by developing a training activity matrix covering the full career span of a trial statistician.
If you’d like a copy of the checklist or wish to share feedback from piloting, please contact the Secretariat at regctus@leeds.ac.uk.
Additionally, we are collating a list of training materials which CTUs have developed or use with their new statisticians. Our aim is to create a repository which network colleagues can access and offer up new materials for. Seven CTUs responded to us initially but we are still keen to hear from a wider range of units and invite interested colleagues in other CTUs to become involved.
Annual National Meeting
Plans are underway for the upcoming annual national statistics meeting, hosted by King’s CTU, to be held on 13 May 2026 in Central London. Details to follow soon.
– Stephen Bremner (King’s CTU) & Jo Haviland (Pragmatic CTU)
Monitoring Group Update
Monitoring Clinics
70 participants joined in February 2026 for a discussion about the new clinical trials regulations as they effect clinical trial monitoring. Thank you to everyone who took part.
Further sessions are planned for 2026, so ask your CTU monitoring lead for the TEAMS link if you would like to join in the conversation. You can also email s.love@ucl.ac.uk if you would like a particular subject to be covered in future clinics.
Save the date for the clinical trial monitoring annual meeting on 18 June 2026
The annual clinical trial monitoring meeting will take place on 18 June 2026 as a virtual meeting. The agenda will be circulated soon. Please hold this date in the meantime.
Central data monitoring of clinical trials
Thank you to all of those who took part in our survey on central data monitoring. This enabled us to identify five key recommendations. This has been accepted for publication and the proofs submitted. More information on this to follow in due course.
Greener monitoring case studies
The Greener monitoring group have developed some case studies to help with remote monitoring. These will be released shortly.
Do contact the UKCRC network at rectus@leeds.ac.uk if you want to become actively involved in any monitoring projects.
– Sharon Love (UCL Innovative CTU)
Chief Investigators Network Group (CING) Update
Ongoing Activities
CING continues to work on its programme of activities to further strengthen the collaborations between a chief investigator (CI) and a CTU.
I am extremely grateful to Prof Katie Morris (Birmingham CTU) for spearheading our update of the Network’s Checklist for Chief Investigator Engagement. This document was last released in February 2018. Katie has used a combination of AI and real live human beings to create, review, and finalise an updated template for a Guide to CI and CTU Collaboration. We are grateful to Edinburgh CTU for sharing some materials to help us with this. After internal approvals, we anticipate releasing this in time for the next CTU Directors’ meeting via https://ukcrc-ctu.org.uk/chief-investigators/.
Meantime, we are developing a blueprint for an Associate CI Scheme and an Educational Resource for CIs, whilst navigating our new status as the seventh Operations Group in the Network.
Training for Chief Investigators
Finally, after a record attendance at QMUL’s How to be a Great CI Course in January, we are looking forward to the next course in Nottingham on 5 May and in Birmingham on 28 September 2026.
PLEASE TELL YOUR NEW CIs to register here for the course in May: https://ukcrc-ctu.org.uk/how-to-be-a-great-chief-investigator-for-clinical-trials-one-day-workshop/.
– Rustam Al-Shahi Salman (Edinburgh CTU)

Training & Development Opportunities
The Network, and its member CTUs, are committed to sharing their knowledge of best practice in clinical trials research from the absolute fundamentals of trial development and management to the use of the latest novel trial designs.
You can find out about some of the upcoming learning and development opportunities taking place across the Network here.

Job Opportunities Across the Network
Working in an an academic or non-commercial CTU can offer you exciting opportunities that may not be available to you elsewhere. These range from publishing your research, presenting at national or international conferences, or teaching the next generation of researchers, as well as making a positive difference to patient and public health.
You can find out more about current vacancies across the Network here.
THE INFORMATION INCLUDED BELOW HAS BEEN PROVIDED BY PARTNER ORGANISATIONS
AND IS INCLUDED WITHOUT EDITS

UKTMN Update
UKTMN Annual Conference 2026
The UKTMN Annual Conference is scheduled to take place in Cardiff on 9 June 2026. Registration, open to UKTMN members, opened in January. A call for abstracts was also announced earlier this year, which closed on 20 March 2026. All submitted abstracts are now undergoing review and an updated programme will be released shortly after decisions are shared at the end of April.
The programme includes keynote presentations from Alice Mortlock (DHSC) and Paula Williamson (TMRP), parallel sessions, trial management presentations, posters, and networking. Find out more here.
UKTMN ASPIRE leadership programme
The ASPIRE leadership programme has been specifically tailored for trial management professionals. Cohort 2 (pictured right) completed the six-month programme in February 2026 with a closing session in Manchester.
Applications for Cohort 3 are now open to UKTMN members, with a start date of October 2026.
Learn more about the programme, and how to apply here.

UKTMN training
Several UKTMN training courses are scheduled to take place in the next few months, including How to be a good trial manager (fully subscribed), an Introduction to grant writing for trial management (fully subscribed) and Recruitment and retention (places remaining). The UKTMN training group are also working on addressing gaps in current training provision in line with findings from a training survey completed by members, with some brand-new courses in the pipeline.
All training events available for registration can be found on the UKTMN Event list.
UKTMN Webinars
There are several upcoming UKTMN webinars available for registration, including ‘Future-Proofing Clinical Research: Durable Infrastructure and Platform Trials and NIHR- Be part of Research, with previous webinar recordings also available to catch-up on, including ‘applying burnout theory to trial management’ and ‘Reaching the Right Participants- How Paid Social Media Advertising Accelerates Study Recruitment’.
Recordings for all past UKTMN webinars can be found here.
DigiTrials Update
Datasets Survey
We are doing a piece of work to better understand the datasets DigiTrials uses within its services, as well as which additional datasets would be most valuable to onboard in the future. As part of this work, we are seeking to recruit participants to complete a short survey.
We are looking to hear from people who have experience in:
– Assessing the feasibility of clinical trials, and/or
– Recruiting participants for clinical trials or health research studies
The survey is currently open and will close on 24 April 2026. It should take approximately 15 minutes to complete.
If you would like to take part, you can complete the survey here.
Thousands recruited for a “new era” severe mental illness study
NHS England’s DigiTrials service is powering a landmark moment in mental health research, helping to identify and recruit up to almost 50,000 eligible participants for GlobalMinds – the world’s largest severe mental illness study. This groundbreaking programme is accelerating world‑leading clinical research and bringing the promise of personalised treatment pathways closer than ever before.

The three‑year study, led by Akrivia Health – a University of Oxford and NHS spin‑out – will link genetic data with questionnaire responses and NHS medical records to create the most detailed dataset ever compiled on severe mental health conditions. This transformative approach will deepen our understanding of bipolar disorder, schizophrenia, psychosis and major depression, opening the door to earlier diagnosis, more precise treatments, and better outcomes for patients.
Akrivia’s platform already supports more than 6.3 million patients across 20 NHS organisations by turning unstructured clinical information into research‑ready insights. GlobalMinds builds on this foundation by adding genomic data, marking a major step forward in how we understand and treat serious mental illness. Learn more.

NIHR Update
Masters for Research Delivery Leadership: Bursary applications now open
Are you a health or care professional with a passion for improving patient care?
Bursary applications are now open for Masters for Research Delivery Leadership – a range of masters level qualifications that will give you the skills to take on leadership roles in research delivery.
The part-time courses consist of practical work-based learning and online modules designed to fit around your busy work schedule.
Applications close midday, 1 July. Find out more and apply on the NIHR website.
Now Open – Applications Open for NIHR Developing Research Leader Programme
Are you a nurse, midwife or allied health professional passionate about developing your research leadership and embedding research in health and care?
We’re pleased to announce that applications are now open for the NIHR Developing Research Leader (DRL) Programme.
The DRL Programme supports nurses, midwives and allied health professionals to grow as research leaders, champion research within their organisations, and help build a sustainable and inclusive research culture across health and care settings.
The 3-year programme provides protected time, structured development and mentorship, and access to a national network of peers – enabling participants to strengthen their leadership skills and increase research capacity in practice.
Applications close on 21 April 2026. Find out more and apply on the NIHR website.
Improving information governance (IG) review processes
Tees, Esk and Wear Valleys NHS Foundation Trust has carried out a project to speed up the information governance (IG) review process for health research, resulting in faster study set-up times.
Driven by the UK government’s goal of reducing clinical trial set-up to under 150 days, the project addressed a key delay: the common practice of research sites duplicating the national IG review already completed by the Health Research Authority (HRA).
Colleagues from the trust’s research and development team were supported by the NIHR RDN to develop the new process. Now, studies not sponsored by the trust and undergoing HRA’s study-wide review will no longer be routinely submitted for a duplicate local IG check. This clarity reduces risk from conflicting assessments and has already led to faster set-up for complex commercial studies.
The HRA is now incorporating these – and other – system learnings into its new ‘What Good IG Looks Like’ guide, helping other trusts nationwide adopt a more proportionate and efficient approach.
NIHR support for sites to accelerate study set-up times
Research sites can now access a range of standard and enhanced support via the NIHR Research Delivery Network to reduce study set-up times to under 150 days.
Watch a new video on the NIHR’s 150-day webpage to find out more.
Update: Evolving the Participant in Research Experience Survey (PRES); Launch of ‘My Research Experience’
As you may be aware, the NIHR undertakes a national survey that asks adults and children what they think and feel about taking part in research supported by the NIHR. This has been known as the Participant in Research Experience Survey (PRES).
The National Institute for Health and Care Research (NIHR) is implementing significant updates to the Participant in Research Experience Survey (PRES), effective from April 1st, 2026, renaming the survey to ‘My Research Experience‘. This will help to enhance public awareness, research team engagement and will align the survey with the digital platform myresearchexperience.com.
Alongside the change in name, an updated adult question set will be launched following a collaborative review. As part of the updates, there will be a national communications campaign called, ‘Every Contact Counts‘ launching on 1 April until early summer 2026, informing staff of the key updates. An emphasis on being able to offer the survey more than once during a study’s lifetime and the eligibility of all NIHR portfolio studies for My Research Experience will be a key feature of this campaign.
We are pleased to share these important updates with you as key stakeholders and partners and we want to reassure you that the aim of the survey, the core delivery processes, accessibility options and data reporting timelines, will remain unchanged.
We would be more than welcome to discuss any questions you may have about My Research Experience. Please consult the FAQ guidance or alternatively, you can reach out to our Regional Research Engagement and Inclusion Teams or the national Public Partnerships team at rdncc.ppt@leeds.ac.uk. Information for Delivery Organisation colleagues and external stakeholders can be found on the RDN website.
Protect and improve your wellbeing
NIHR has released its latest e-learning module to help you build the self-awareness, skills and practical strategies needed to protect and improve your wellbeing.
The 40-minute module will support you to strengthen your personal resilience, set healthy boundaries, practice self-compassion and foster meaningful connections.
➡️ Find out more on NIHR Learn.
Build a high performing team
Assess where your team is, reflect on what your best looks like and discover strategies for managing conflict by watching NIHR’s latest webinar. It also explores how to embed true inclusion and build psychological safety.
Compassionate and inclusive leadership
Cultivate a nurturing environment by engaging and empowering others to flourish. Attend our webinar on 1 June from 1-2pm to understand why compassionate leadership and inclusivity matters.
It will share what the evidence says about their impact in health and social care settings and explore 4 habits for more inclusive, everyday leadership. Find out more.
New AcoRD Training Course Launched
The new AcoRD training course is now live and available in NIHR Learn.
The attributing the costs of health and social care research and development (AcoRD) guidance provides a framework for the NHS and its partners to identify, attribute and recover the costs of health and social care research and development in a transparent and consistent way.
Who:
This course is aimed at Study Representatives (researchers and Sponsors) and AcoRD Specialists but it is open access and can be shared with anyone with an interest, i.e. wider R&D community, funders, RDN staff.
What:
Module 1: Background and Principles
The principles of the AcoRD guidance, how it fits into the research landscape, the different cost attributions, how to identify and attribute different study activities and where you can find additional support.
Module 2: Tools
The tools available to support the cost attribution of study activities, such as the Schedule of Events Cost Attribution Tool (SoECAT), the process for SoECAT completion and authorisation, the Sponsor’s role and the consequences of poor attribution.
Module 3: Authorisation
The benefits and importance of Schedule of Events Cost Attribution Tool (SoECAT) authorisation and how to escalate any attribution queries (aimed predominantly at AcoRD Specialists).
Module 4: Commissioning and Excess Treatment Costs
The importance of commissioners, the commissioning process and how this relates to Excess Treatment Cost payments.
Additional resources: NotebookLM
A view-only, AI-powered learning aid designed to help users interact with the course materials, summarise key concepts, and answer specific questions based on the course content.
Where:
The AcoRD training course can be accessed from NIHR Learn and it is open (self-enrollment). Please note, users will need to register for an NIHR Learn account to enrol onto the module. NIHR Learn itself is available to NHS, UK universities, and other publicly funded organisations conducting and supporting health and care research.
Why:
This course has been developed to provide a more rounded, centralised and accessible resource for use by all. It replaces the original DHSC slideset and training we used to run via the R&D Forum and is intended to complement any training provided by Study Support Service colleagues in RRDNs/AcoRD Specialists across the UK.
Please contact the Triage Helpdesk at: triagehelpdesk@nihr.ac.uk with any questions or feedback.
Further resources in this space:
~ What is AcoRD?
~ Online SoECAT Guidance Page
~ Online SoECAT Guidance Module
~ Excess Treatment Costs







